MACULAR DEGENERATION
How Common is Macular Degeneration?
Macular degeneration, also known as age-related macular degeneration, AMD or ARMD, is a sight-threatening eye condition. It is responsible for 50% of legal blindness in Australia.
1.9 million Australians currently live with macular degeneration. One in seven Australians over 50 years of age show signs of macular degeneration. A staggering 8.5 million Australians are at risk of developing macular degeneration. 288 million people world-wide will be affected by 2040.

What are the Different Stages of Macular Degeneration?
There are different stages of macular degeneration based on clinical signs: early, intermediate and late.
In the early stage, your vision is usually normal. Your eye specialist may have noted some drusen, pigment changes at the macula when they perform an OCT scan. It is important at this stage that you are monitored routinely. You should also self-monitor at home for any changes in your vision.
Intermediate and Late Wet (Neovascular) Stage
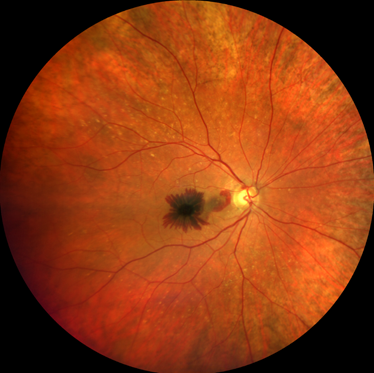
In intermediate and late stages, vision can change rapidly. Leakage and bleeding of the abnormal blood vessels under the retina (called choroidal new vessels) can occur and they can cause a sudden loss of vision or distortion in your vision. This is called wet or neovascular macular degeneration and it requires prompt assessment and treatment.
Alternatively, in the late stage, the retina may become thinned (atrophic) with loss of photoreceptors at the macula. This is the late dry type of macular degeneration. You may notice gaps in your vision (called scotoma) and changes usually occur gradually. However, your vision can change rapidly, if you already have advanced macular degeneration in the other eye or if there is a family history.
What is the Best Treatment?
Intravitreal injections for wet (neovacular) macular degeneration
Current treatments for wet (neovascular) macular degeneration include intravitreal injections with anti-VEGF drugs such as Eylea (aflibercept) and Eylea HD. These drugs treat macular degeneration by inhibiting the growth of abnormal blood vessels and help prevent further leakage or bleeding. Many of our patients have successfully stabilized their vision for many years, with comprehensive follow-ups and regular injections.
New treatment for geographic atrophy
For the first time, there is treatment available for geographic atrophy, the late stage dry type of macular degeneration. Syfovre (pegcetacoplan) has been approved by the TGA for treatment of geographic atrophy.
Izervay (avacincaptad pegol) is another new treatment on the horizon for geographic atrophy.
What is an Intravitreal Eye Injection?
Eye injections are performed as an out-patient procedure at our practice with topical and local anaesthetic. Dr Lawrence Lee has had many years of experience providing these treatments. Our team will look after you and make you feel at ease and as comfortable as possible during the procedure. There is minimal pain. After the injection, Dr Lee will prescribe you with antibiotic eye drops to help prevent infections. We also advise that you keep your eye clean and contact us immediately if you notice any redness, pain or changes in your vision.
Many of our patient have successfully been able to maintain their vision with regular treatment. How often you need to have injections depends on the treatment plan tailored by Dr Lee. With the new Eylea HD, some patients are able to maintain good vision with an injection every 3-4 months.
Do you have questions or concerns about your eye health or a specific eye condition? To discuss, please contact City Eye Centre for more information.
