A Milestone: New Geographic Atrophy Treatment — Syfovre®
Geographic atrophy (GA) is an advanced, late-stage form of age-related macular degeneration (AMD). AMD is the leading cause of progressive, irreversible loss of central vision in older adults. GA is sometimes called late “dry” AMD because it doesn’t involve leaking blood vessels. This is unlike the intermediate and late “wet” stage of AMD. Rather, it involves the slow degeneration of retinal cells, particularly the photoreceptors and retinal pigment epithelium (RPE). As these cells die, discrete patches of atrophy progress and expand across the macula. As a result, this leads to worsening central vision over time.
If you have been diagnosed with GA, you may initially notice minor difficulties with reading or recognising faces. However, over time the loss can become profound, severely impacting your independence, mobility, and quality of life.

What Is Syfovre®? Why is it a Milestone?
Syfovre® (generic name: pegcetacoplan) is a groundbreaking treatment that became the first therapy approved to slow the progression of GA.
Originally approved in the United States in 2023, Syfovre received regulatory approval in Australia in January 2025, becoming the first ever approved treatment for GA. Prior to this, there were no treatments to slow or alter the course of GA.
Syfovre’s approval represents a paradigm shift in GA care because for the first time, there is a therapeutic new geographic atrophy treatment that can alter the course of this once untreatable disease. By intervening in the immune-mediated process of retinal degeneration, we can now offer a treatment. This is aimed at effectively slowing down the progression of the lesion and helping preserve vision.
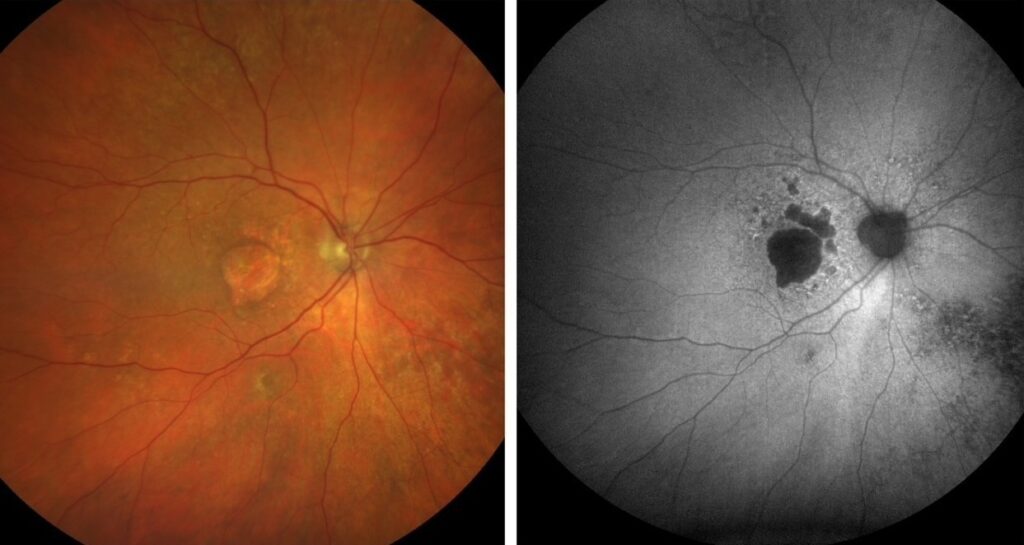
How Does Syfovre® Work?
Syfovre® targets part of the immune system’s complement pathway — a cascade of proteins involved in inflammation. When overactivated in the retina, this contributes to GA lesion growth and vision loss.
It is administered via intravitreal injection, typically given on a schedule of every other month.
Is Syfovre® Safe and Effective?
The efficacy of Syfovre® were established in large clinical trials. These studies showed that:
- Syfovre® significantly reduced the rate of GA lesion growth
- The treatment effect increased over time, with greater reductions observed at 18–24 months.
- Long-term data demonstrated sustained benefits over 3 to 5 years. One analysis showed that lesion progression was delayed by roughly 1.5 years in treated patients versus controls.
Syfovre®’s safety profile has also been well characterised in trials and real-world use. The most commonly reported side effects include mild inflammation, conjunctival haemorrhage, and floaters. The treatment is generally considered tolerable, and its benefit in slowing disease progression has made it a valuable option for eligible patients.
Can patients get access to this treatment now?
Geographic atrophy has long been a leading cause of irreversible vision loss with no effective therapies. The arrival of SYFOVRE® and other complement-targeted drugs has fundamentally changed the landscape by offering the first strategy to slow disease progression.
Syfovre® Patient Access Program commences on 2 March 2026. For more information, please contact us at City Eye Centre on 07 3831 6888 or email us at appointments@cityeye.com.au.
